Current Research
Dendritic Cell Development And Function
Novel regulators of DC differentiation
We have adopted an in vitro system of DC differentiation for genome-wide CRISPR/Cas9 dropout screening. Using this approach, we have identified multiple novel regulators of the differentiation of DCs and related cell types such as macrophages. Study of these factors and their target genes should help us understand the mechanisms of DC and macrophage specification in the steady state, inflammation and cancer. Finally, we are characterizing the role of chromatin organization in the differentiation and function of DCs.
Dendritic cells (DCs) are key sentinel cells of the immune system that directly recognize pathogens and initiate adaptive T and B cell responses by secreting cytokines and presenting antigens to T cells. The DC lineage includes interferon-producing plasmacytoid DCs (pDCs) and antigen-presenting conventional or classical DCs (cDCs), the latter comprising at least two genetically and functionally distinct subsets (cDC1 and cDC2). In addition to their essential role in antimicrobial and antitumor immunity, DCs control immune homeostasis, tolerance and autoimmunity. Indeed, aberrant activation of DCs has been implicated in nearly all autoimmune diseases.
A major goal of the lab is to characterize the molecular control of DC development and function.
Mechanisms Of Systemic Autoimmunity
Immune System Development In Unmanipulated Animals
Pathogenic T cells in SLE
Origin and significance of anti-D1L3 antibodies in SLE
Plasmacytoid dendritic cell function
Dendritic cell in human tissues
pDCs are capable of rapid and massive secretion of interferon type I (IFN-I) in response to viruses. Although they manifest multiple adaptations for this role, such as secretory morphology and the expression of endosomal Toll-like receptors and IRF7, these features are neither unique to pDCs nor fully explain their functionality. The goal of the lab's ongoing studies is to characterize the mechanism of virus recognition and IFN-I production by pDCs.
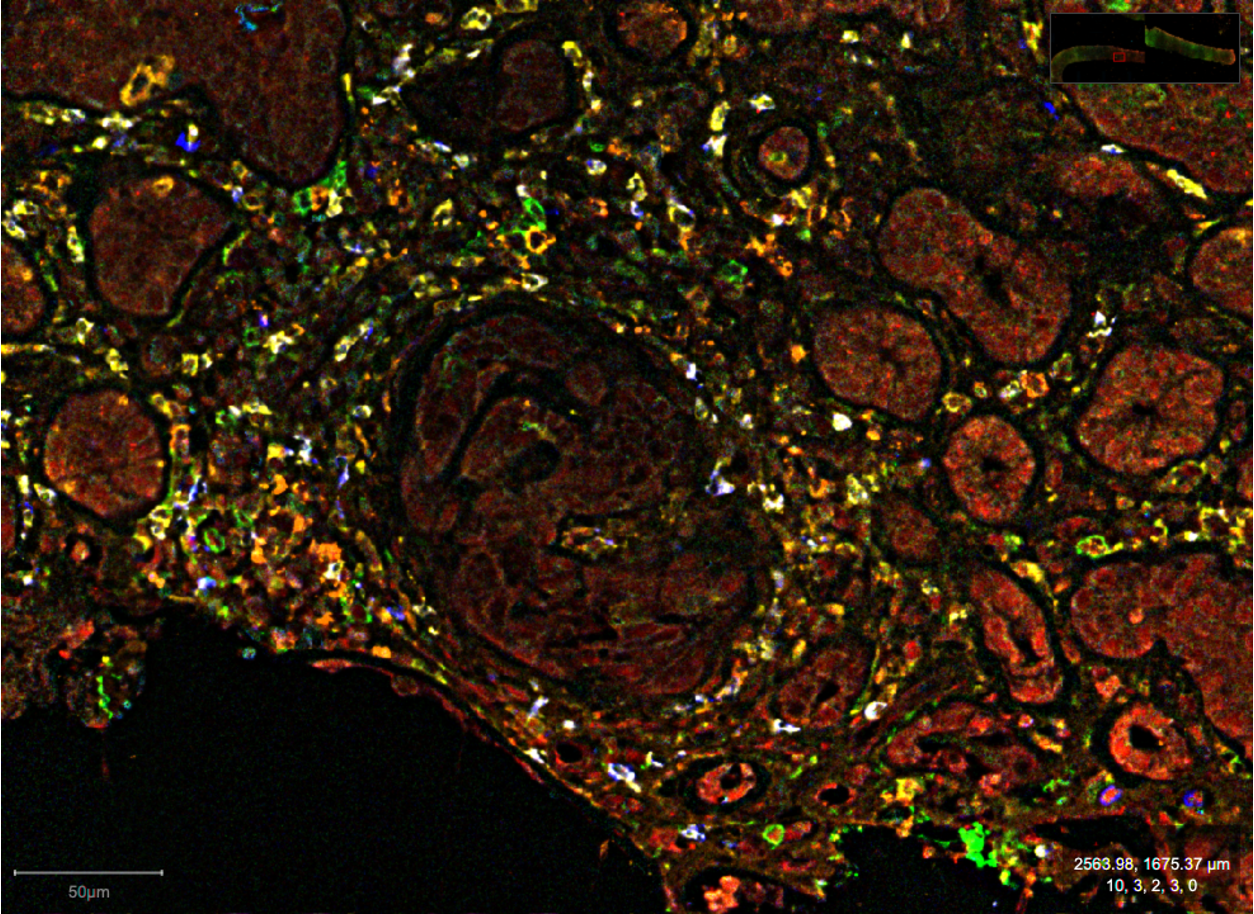
We have been using high-dimensional immunochemistry in order to visualize myeloid cells including DC subsets in murine and human tissues. We are analyzing the spatial distribution and interactions of DCs in the lymphoid organs and tissues from from human organ donors, as well as from patients with various cancers.
Antibodies to chromatin and DNA are a hallmark of autoimmune disease systemic lupus erythematosus (SLE) and a harbinger of its severe complications such as lupus nephritis (LN).
A major goal of the lab is to characterize the mechanism of tolerance to self-DNA and its subversion in systemic autoimmune diseases such as SLE. To this end, we are focusing on DNASE1L3, a secreted DNase whose absence causes monogenic SLE in human patients.
Pathogenesis of lupus nephritis
Hematopoietic abnormalities in aging and cancer
Hematopoietic abnormalities in SLE
T cells are essential for anti-DNA responses in SLE, but the repertoire and specificity of T pathogenic helper T cells in these autoimmune responses are unknown. We are using DNASE1L3-deficient mice to identify T cells that facilitate anti-DNA responses, and to characterize their repertoire and specificity.
Anti-DNA autoreactivity leads to severe SLE complications such as lupus nephritis, but the mechanism of transition from preclinical autoimmunity to overt nephritis remain poorly understood. We are using DNASE1L3-deficient mice to study genetic and environmental factors that may induce the transition to overt disease.
We discovered that many SLE patients with LN have reduced DNASE1L3 activity, which is associated with the presence of neutralizing Abs to DNASE1L3. We are using sequencing approaches to clone these antibodies from patients and characterize their affinity, epitope specificity and cross-reactivity with other self-antigens.
Dendritic cells and other cell types in the immune system (e.g. granulocytes and lymphocytes) and the blood (e.g. platelets and erythrocytes) are being continuously generated in the bone marrow throughout the entire lifespan. At the apex of this process are self-renewing, multipotent hematopoietic stem cells (HSCs) that can reconstitute the entire hematopoietic system upon transplantation into irradiated hosts.
A major goal of the lab is to understand the development of immune cells from HSCs in unmanipulated animals, i.e. without transplantation.
We have previously used lineage tracing to characterize the rate and kinetics of blood cell development from endogenous HSCs in the steady state. We are currently using this approach to characterize the effect of aging and malignant transformation on HSC-driven hematopoiesis.
Hematopoietic abnormalities such as thrombocytopenia and lymphopenia are widespread and often life-threatening in autoimmune diseases such as SLE, yet little is known about their pathogenesis. We are examining the cross-talk between systemic autoimmunity and HSC-driven immune cell development, and the potential role of stem/progenitor cell abnormalities in SLE.